
Therefore, to harmonize the approach between GAMP. 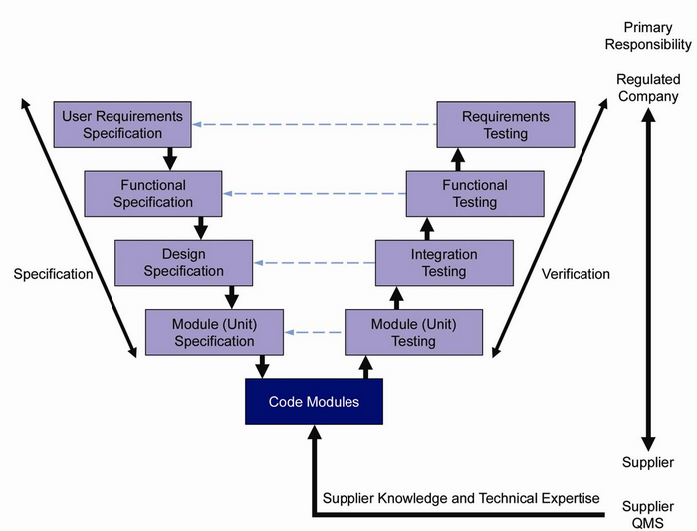
a single document to accomplish the complete validation. The Guide facilitates the effective and efficient use of valuable resources by the application of appropriate and proportionate practices, encouraging innovative approaches to managing risk to patient safety, product quality, and data integrity, while supporting benefit to public health. based on a risk-based approach for category 3 software using. Technological innovation is essential for life sciences industries in providing value to society while also controlling costs and reducing time to market. The ISPE GAMP ® 5 Guide: A Risk-Based Approach to Compliant GxP Computerized Systems Second Edition aims to protect patient safety, product quality, and data integrity by facilitating and encouraging the achievement of computerized systems that are effective, reliable, and of high quality. Other examples that would fit under GAMP category 3 would be systems that WebCategory of software. GAMP ® guidance does not define a prescriptive method or a standard, but rather provides pragmatic guidance, approaches, and tools for the practitioner. GAMP Software Category 1 Infrastructure Software. Promotes a system life cycle approach based on good practice.Category 4 Configured software including, LIMS, SCADA, DCS, CDS, etc. Establishes a common language and terminology Category 3 Non configurable software including, commercial off the shelf software (COTS), Laboratory Instruments / Software.Therefore, to harmonize the approach between GAMP ®. Facilitates the interpretation of regulatory requirements based on a risk-based approach for category 3 software using. V Model Computer System Validation GAMP 5 CSV V Shaped Model for CSVIn this video I discussed one type of methodology used for validation projects.GAMP® is an ISPE Community of Practice (CoP). Using the GAMP5 software categorization, laboratory computerized systems fit into software categories 3 and 4 and to some degree Category 5, although it needs to be noted that Categories 3 to 5 are effectively a continuum with no absolute boundaries.10The categories are defined as follows: Category 3 Non-Configured Commercial Products: this. 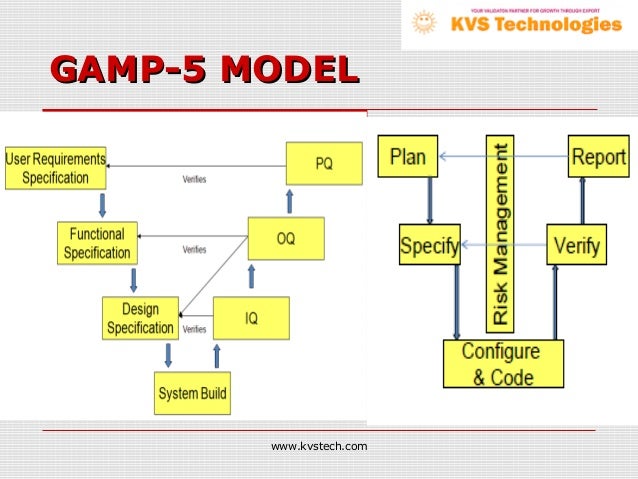
GAMP® adopts a patient-centric risk-based approach that enables innovation while demonstrating compliance with regulatory requirements. The overall approach, framework, and key concepts remain unchanged from the first edition. It aims to achieve computerized systems that are fit for intended use and meet current regulatory requirements by building upon existing industry good practice in an efficient and effective manner. GAMP 5 Guide, 2nd Edition, aims to continue to protect patient safety, product quality, and data integrity by facilitating and encouraging the achievement of computerized systems that are effective, reliable, and of high quality. GAMP ® guidance aims to safeguard patient safety, product quality, and data integrity in the use of GxP computerized systems.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
